

The Diamond-Anvil Cell
The diamond-anvil cell is a simple device capable of generating pressures almost as great as those found on the centre of the Earth. By applying these pressures to small samples we can then study how materials behave under high presures by a variety of techniques.
Pressure is defined as the force applied to a given area. Thus, to increase pressure one can either increase the force being applied, or one can decrease the area.
High pressures are generated in the diamond-anvil cell by taking a small force applied to a large area (ie low pressure) and transferring that same force to a small area.
Pressure is defined as the force applied to a given area. Thus, to increase pressure one can either increase the force being applied, or one can decrease the area.
High pressures are generated in the diamond-anvil cell by taking a small force applied to a large area (ie low pressure) and transferring that same force to a small area.
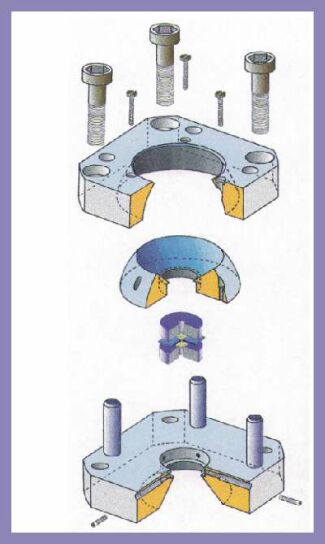 |
This is an exploded view of the diamond-anvil cell that was developed in 1996 at the Bayerisches Geoinstitut in Bayreuth by Allan, Miletich
& Angel (Rev. Sci. Instrum. 67, 840-842).
Force is generated by the four screws which, when tightened, draw the two halves of the cell together. This force is then transmitted through the steel body of the cell, through the beryllium plates to the diamond-anvils. 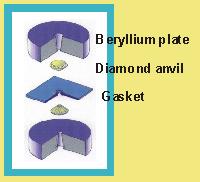
The two anvils are brilliant-cut diamonds, almost the same as found in wedding rings except that small flat surfaces are ground on their tips. High pressure is generated because the area of the diamond tips or culets is much smaller than the area of the screw threads. |
 |
These are the parts of the ETH diamond-anvil cell developed from the BGI design by Ronald Miletich. There are mechanisms for adjusting the two anvils so that they are exactly parallel and aligned on-axis with each other. Without alignment the diamonds would cut through the gasket and destroy the sample. Alignment between the two halves of the cell is maintained by the four pins that are fixed into one half of the cell and slide through the holes in the other half. The four drive screws are also shown alongside a US quarter coin. |
|
Why diamonds? In addition, the diamonds are transparent to many radiations including light (so that we can see the sample inside the cell) and X-rays. |
Why beryllium? |
| The sample crystal is placed inside a small hole, 0.25mm diameter, that is drilled in a thin metal foil. This metal foil gasket serves two purposes. It keeps the diamonds from crushing the crystal. And it allows us to surround the crystal with a fluid that provides hydrostatic conditions for the crystal. |  |
This is a view through the diamonds into the high-pressure chamber formed by the gasket.
The red crystal is the sample, the pale crystal is a quartz crystal used for precise pressure measurement, and the spheres are ruby crystals used for approximate pressure measurement. |
 |
This is an assembled ETH diamond-anvil cell. It is mounted on a goniometer head that attaches it to the diffractometer for X-ray diffraction measurements. |  |
- the way structures change with pressure - the density of minerals at high pressure |
Thanks to Ronald Miletich (ETH Zurich) for the drawings on this page and the picture of the crystal in the cell. Mark Fortney (VPI) took the other photos.